FluoroType® MRSA – Fast, easy and reliable
Your test system for direct detection of methicillin-resistant Staphylococcus aureus strains from patient samples
With FluoroType® MRSA we offer you a particularly userfriendly and efficient solution for the fast detection of MRSA directly from patient samples: The test system enables reliable results within only 1.5h by using highly specific fluorescence-labelled probes.
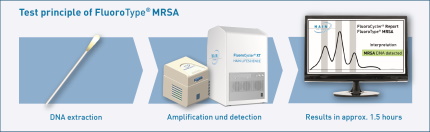
The test is performaned directly from clinical swabs and requires only minimal pipetting effort. From amplification to detection, all steps run fully automated in the FluoroCycler® 12 or the FluoroCycler® XT. A ready-to-use amplification mix already containing the Taq polymerase is provided with each kit. Therefore, integration of the FluoroType® MRSA into labs with small, medium and large throughput is easily possible. The result is interpreted by the test-specific FluoroSoftware® and is visible at first glance. FluoroType® MRSA gives you the crucial time and cost advantage for reliable and efficient MRSA diagnostics.
Your benefits with FluoroType® MRSA
- Fast: Swabs from nose, throat, skin and wounds can directly be used for the simple test procedure. Results are available after only 1.5 hours.
- Reliable: Dropout strains are identified via the simultaneous detection of the SCCmec cassette and the resistance-mediating mecA gene. The additional detection of the gene homologue mecC makes the results even more reliable. An Internal Control ensures valid results from sample preparation to amplification and detection.
- User-friendly: From amplification to result, all steps run fully automated in the FluoroCycler® 12 or the FluoroCycler® XT. A ready-to-use amplification mix containing the Taq polymerase is provided with each kit. Therefore, FluoroType® MRSA can easily be integrated into your daily lab routine.
- Flexible: With FluoroType® MRSA single samples as well as big batches can be analysed efficiently.
At a glance
Molecular genetic assay for automated detection of methicillin-resistant S. aureus strains from patient specimens
Starting material:
Swab specimens (nose, throat, skin and wounds)
Swab:
transystem® 108C
DNA Isolation:
SpheroLyse
Instrument for Amplification and Detection:
FluoroCycler® 12
Order number:
96 Tests No. 62596
FluoroType® technology
Downloads:
Information request:
More Information: